2. Van der Waals is another weak force involving the interaction between the permanent diploes of two uncharged polarized bonds (dipole-dipole) or the interactions between a permanent dipole and a transient dipole induced in a neighboring molecule (instantaneous dipole-induced dipole).
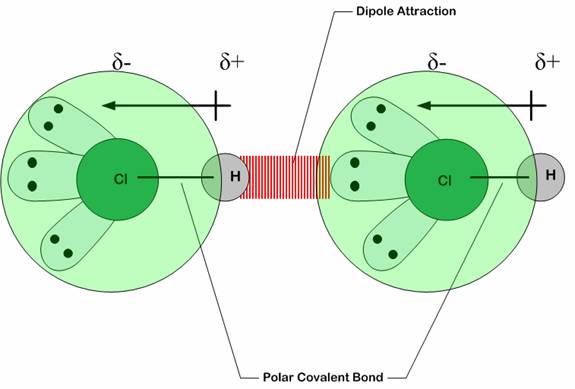
In dipole-dipole interactions, both the molecules are uncharged yet polarized, meaning they are not ions with an extra or missing electron (anion or cation), rather, they are molecules with one end being more electropositive and another more electronegative. Such as Hydrochloric acid (HCl). When two HCl come close, they immediately configure in such a way as to have their opposite poles face each other.
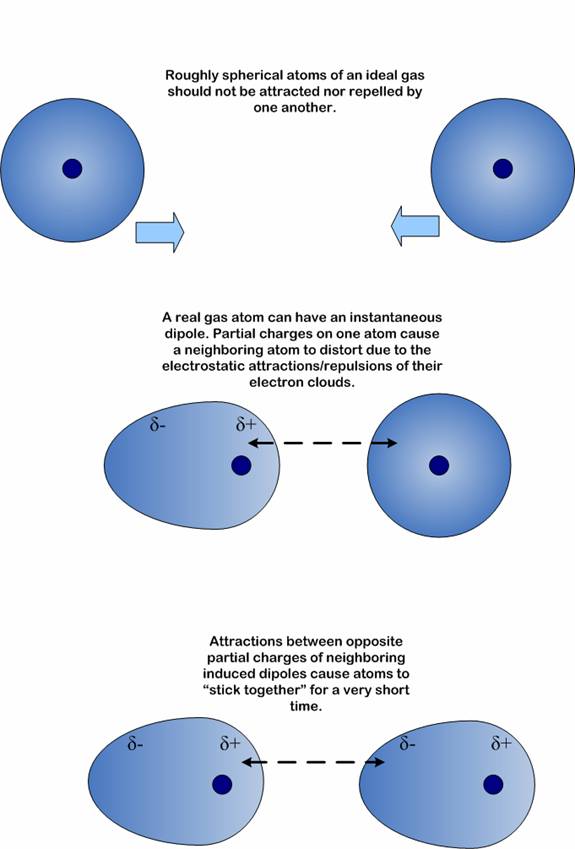
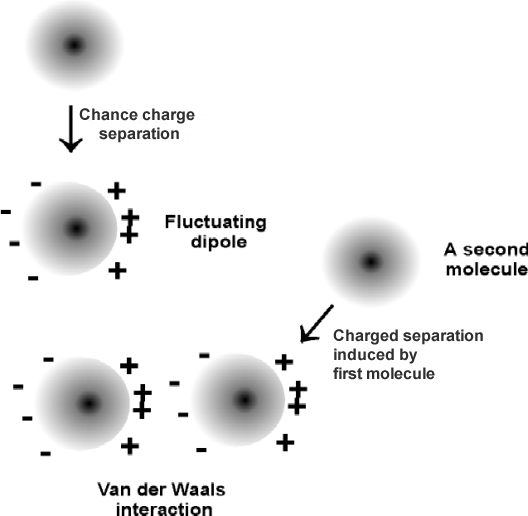
In instantaneous dipole-induced dipole, the atom’s or molecules’ electrons are in spontaneous presence around the electron orbital causing fluctuations in their polarity structure. Once a dipole is spontaneously created, the incident will induce a polarity in another neighboring molecule.
These forces are of short range and small magnitude.
The attractive forces, also known as London dispersion forces, originate from the infinitesimal dipole generated in atoms by the random movement of the negatively charged electrons around the positively charged nucleus.
Although they operate over similar distances, van der Waals forces are much weaker then hydrogen bonds.
Van der Waals forces also have a repulsive component. When two nuclei are squeezed together, the electrons in their orbitals repel each other. This repulsion increases exponentially as the atoms are pressed together, and at very close distances it becomes prohibitive.
The sum of the attractive and repulsive components of van der Waals forces, are said to be in Van der Waals contact, and the attractive force between them is maximal.
Although individual Van der Waals forces are weak, the clustering of atoms within a protein, nucleic acid, or biological membrane permits the formation of a larger number of these weak interactions.
Once formed, these cumulative weak forces play important roles in maintaining the structure of the molecules.
For example, the heterocyclic bases of nucleic acids are stacked one above another in a double-stranded DNA. This arrangement is stabilized by a variety of noncovalent interactions, especially Van der Waals forces. These forces are collectively known as stacking interactions.