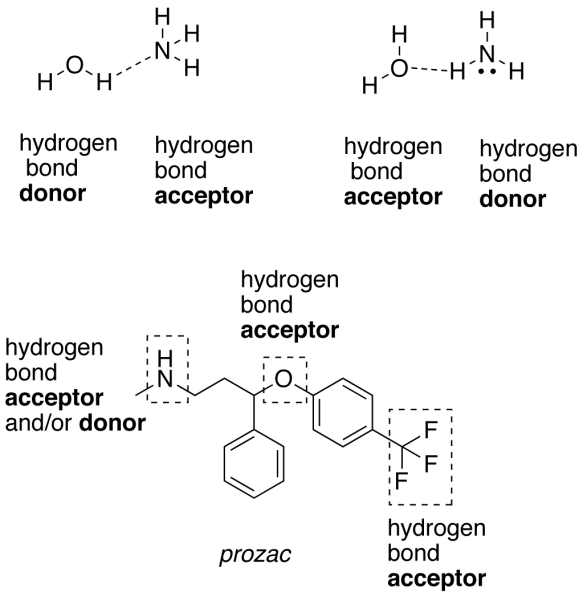
As we’ve already discussed these two before, here is a brief overview again.
3. Hydrogen bonds are among the strongest noncovalent forces in biological systems.
They are paradoxically strong enough to provide structural stability but weak enough to be readily broken.
In general, a hydrogen bond can form when a hydrogen atom covalently bonded to a strongly electronegative atom, such as nitrogen, oxygen, or, in rare cases, sulfur.
Hydrogen lies approximately 0.2 nm from another strongly electronegative atom that has an unshared electron pair. The total distance between the two electronegative atoms participating in a hydrogen bond is typically 0.27 to 0.30 nm. Some common examples of hydrogen bonds are shown.
All the functional groups are also capable of forming hydrogen bonds with water molecules. In order for hydrogen bonds to form between or within biological macromolecules, the donor and acceptor groups have to be shielded from water. In most cases this shielding occurs because the groups are buried in the hydrophobic interior of the macromolecule, where water can’t penetrate.
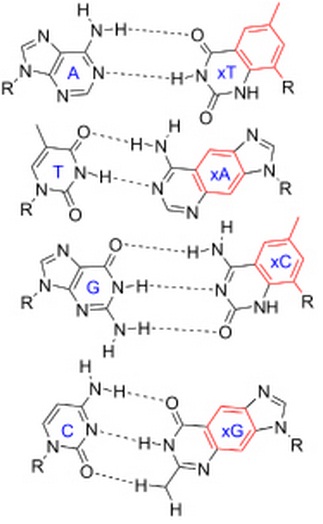
In DNA, for example, the hydrogen bonds between complementary base pairs are in the middle of the double helix as you can see.
4. Hydrophobic interactions: When relatively nonpolar molecule or group in aqueous solution associate with other nonpolar molecules rather than with water, it is termed a hydrophobic interaction.
Although hydrophobic interactions are sometimes called hydrophobic “bonds”, this description is incorrect. Nonpolar molecules or groups tend to group-up not because of mutual attraction but because the polar water molecules around them tend to pressure and entrap them close to each other as the water molecules form hydrogen bonds.
Hydrophobic interactions, like hydrogen bonds, are much weaker than covalent bonds. For example, the energy required to transfer a -CH2– group from a hydrophobic to an aqueous environment is about 3kJ mole-1.
Again, all of the interactions covered here are individually weak compared to covalent bonds, but the combined effect of many such weak interactions can be significant.